Are you interested in our solutions?
We look forward to hearing from you! Feel free to contact us via our contact form.


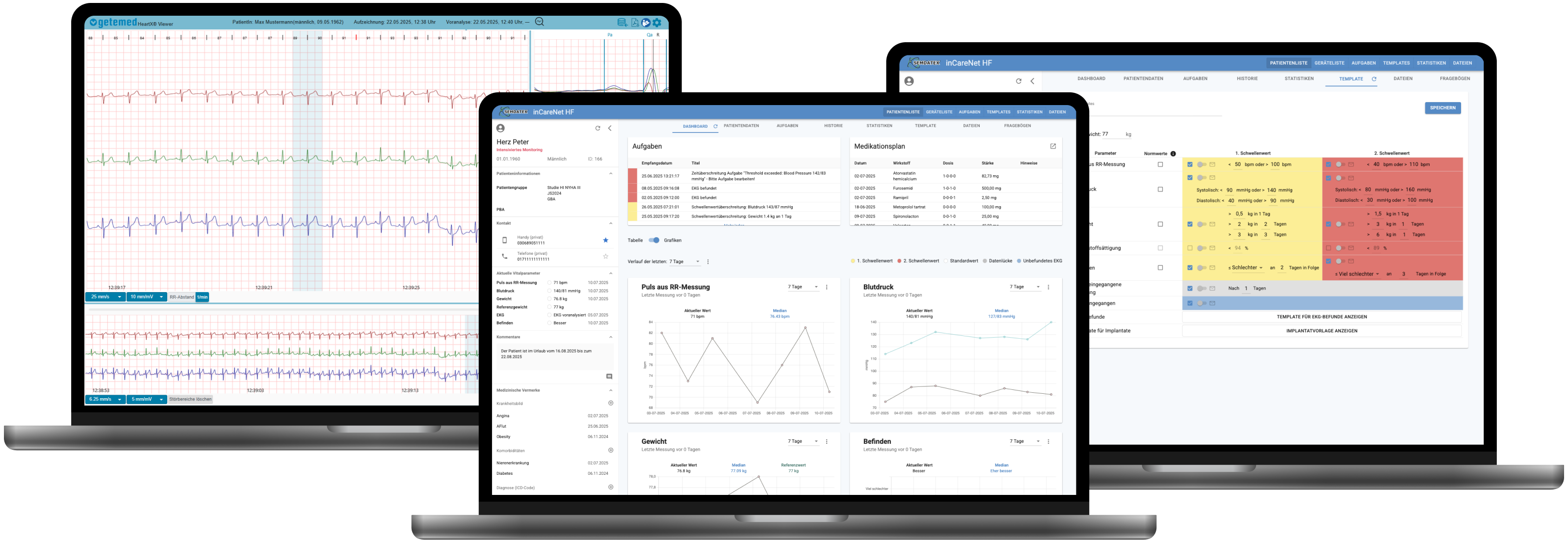
inCareNet HF enables cross-manufacturer monitoring of implants and external devices on a centralized web platform that is certified as a Class IIa medical device.
Individually configurable patient thresholds enable automatic task generation. These tasks can be managed by different network partners across sectors and institutions.
An intuitive, easy-to-read dashboard gives you an instant overview of all relevant cardiac data and medications, helping you stay informed about your patients’ health status at any time.

Our platform meets the highest medical and data protection standards
Our nationwide network of telemonitoring experts provides on-site support — from installation and process optimization to expanding your TMC with network partners and new care modules.
With our solution, you can manage telemonitoring independently – patient data remains in your practice, and medical care stays under your supervision.
Developed by telemonitoring pioneers BIOTRONIK and GETEMED, inCareNet provides a solid foundation for your TMC. By far the most TMC´s in Germany rely on our expertise.
The entire inCareNet HF system is certified as a Class IIa medical device under the EU MDR (Regulation (EU) 2017/745) and complies with German data protection regulations (GDPR).
Most TMCs in Germany rely on inCareNet HF for managing heart failure patients. Your trust is what drives us.
active telemonitoring centres for heart failure patients in Germany
heart failure patients monitored in Germany
Here we explain the most important formal requirements for your participation in the program in Germany and your responsibilities as a telemonitoring center (TMC) operator. Requirements in your country may differ.
Certified specialist in Internal Medicine and Cardiology
Approval for rhythm implant monitoring
Compliance with technical equipment requirements (§ 5 of QS-V TbHI)
Review of incoming alerts and vital parameters on all working days
For intensified monitoring, additionally review alerts on weekends and public holidays
Submit a quarterly report to the Primarily Treating Physician (PTP)
Ensure functioning data transmission
Document the telemonitoring service
Review the indication together with the PTP after 3 and 12 months
Temporary assumption of the PTP function – if needed and in agreement with the PBA


Patient education on implant-based monitoring
We look forward to hearing from you! Feel free to contact us via our contact form.